About CHD-BioCore
Implementing Precision Medicine in Congenital Heart Defects
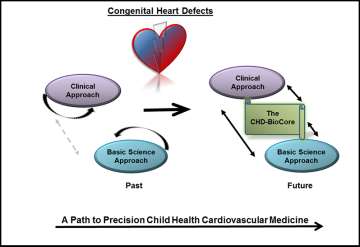
Congenital heart defects (CHDs) affect approximately 1% of live births and are a major source of childhood morbidity and mortality even in countries with advanced healthcare systems. Along with phenotypic heterogeneity, the underlying etiology of CHDs is multifactorial, involving genetic, epigenetic and/or environmental contributors. Clear dissection of the underlying mechanism is a powerful step to establish individualized therapies. However, the majority of CHDs are yet to be clearly diagnosed for the underlying genetic and environmental factors, and even less with effective therapies.
An effective clinical utilization of the new discoveries promises to guide strategic approaches towards improved future outcomes for CHDs but requires narrowing existing gaps between the laboratory bench and the clinic. The importance of dissecting the molecular mechanism of CHDs is at least three folds. First, linking CHDs with specific genetic mutations or environmental exposure can lead to disease prevention, family planning and possible intervention prior to birth. Secondly, better knowledge of the genetic and molecular basis of CHDs would allow personalized approaches to these patients in the prenatal and perioperative settings. Third, with better understanding of gene-gene interaction and gene-environment interaction involved in CHDs, we will be able to identify novel approaches to prevent and effectively treat the pathological manifestation of CHDs. Finally, establishing the CHD-BioCore is essential infrastructure that serves to integrate clinical efforts and research innovation. Our goal is to reduce observations to a focused subset of high specificity on the path to precision child health cardiovascular medicine.
Conclusion
For implementing a path to precision child health cardiovascular medicine, it is important to stratify the phenotypic spectrum of each CHD into sub-cohorts, delineate the distinct genotype associated with each phenotypic sub-cohort utilizing genomic platforms, and establish the pathogenic molecular signature that discriminates the extreme ends of each sub-cohort by implementing RNA-seq. The integration among these key resources by employing systems biology tools is an auxiliary step. An Important infrastructure to achieve these steps requires creation of a continuum from the clinical presentation, hemodynamic indices and physiological parameters, to genomic determinants and gene expression landscape. In order to achieve all these goals, access to high-quality biological specimens from well-defined patient cohorts is a crucial step. Therefore, establishing a CHD-Bio Core is an essential infrastructure and a critical step on the path towards precision child health cardiovascular medicine.