Research Areas
Recent Research
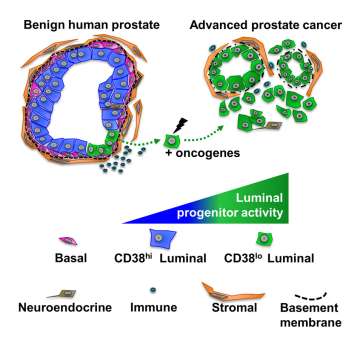
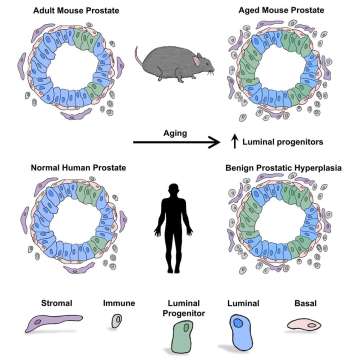
Progenitor Cells and Cancer
Cancer is believed to develop from an accumulation of genetic alterations in normal cells that confer an advantage such as increased oncogenic signaling or loss of tumor suppressors. The cells that acquire these genetic alterations and go on to initiate cancer are termed the cells-of-origin for cancer. Whether there are particular cells in adult tissues that are more likely to develop into cancer in response to genetic alterations has been a question of debate in recent decades. We and others have utilized approaches to distinguish progenitor cells (which can replicate and generate new cells) from differentiated cells and demonstrated that progenitor cells can more easily initiate cancer following the introduction of genetic alterations. As tumors progress and develop resistance to current therapies, tumor cells tend to change their appearance and the genes they turn on or off in order to evade treatments, a process that results in cancer cells more closely resembling progenitor cells. Understanding tumor cell adaptability, and investigating how cancer cells acquire progenitor properties, is important for combating treatment-resistance cancer.
Aging and Cancer
Aging is associated with an accumulation of somatic mutations and epigenetic changes in normal cells. Many cancers are considered diseases of aging. Age is a significant risk factor for prostate cancer, but the mechanisms by which age increases disease risk is poorly understood. Chronic inflammation is associated with aging in many tissues and chronic inflammation is also a risk factor for many age-related cancers including prostate cancer. Mass cytometry has emerged as a tool to interrogate immune populations in dissociated preparations of tissues, and we have utilized mass cytometry for immune profiling in mouse and human prostates. We have demonstrated that luminal progenitor cells are expanded both in prostate tissue with chronic inflammation and in aging prostate tissue. Increasing the number of progenitor cells at risk for transformation likely increases the risk for disease initiation. Understanding how aging alters prostatic inflammation and progenitor content is essential for determining how to prevent or reverse the effects of aging and reduce disease risk.
Metabolism and Cancer
As transformed cells develop into cancer, and cancer cells develop resistance to therapy, their energetic demands change. As tumor cells adapt to a changing environment, the fuels they utilize can also change. Metabolic reprogramming during tumor initiation, progression and treatment-resistance regulates cell survival, proliferation and gene expression. We have found that CD38, a molecule which is highly expressed in prostate luminal cells but absent in aggressive prostate cancer, regulates extracellular NAD+. Determining how cellular metabolism enables tumor cell plasticity is critical for designing effective therapeutic approaches for lethal cancer.