Anthony J. Covarrubias, PhD
Languages
Education
Degrees
Contact Information
Phone
Scientific Interests
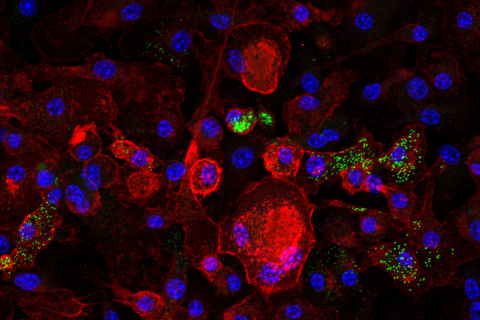
Macrophages are essential immune cells that belong to the innate immune system and are key orchestrators in the initiation and resolution stages of inflammation and can polarize to distinct subsets such as the proinflammatory M1 state or the anti-inflammatory M2 state. Previous studies have shown that chronic inflammation, mediated in part by macrophages, is also a key driver of the pathogenesis of aging-related diseases including cancer. However, the etiology of aging-related inflammation (also known as inflammaging) and how inflammation promotes the development of cancer is not very well understood. Furthermore, upon tumor development, tumor-associated macrophages which reside in high abundance in the tumor microenvironment also play a key role in tumor growth, vascularization, and metastasis. In a recent manuscript Dr. Covarrubias have shown that senescent cells progressively accumulate in aging tissues, leading to activation of resident macrophages to an M1-like state associated with chronic inflammation. These results highlight a new causal link between visceral tissue senescence and tissue inflammation during aging. However, an open question in the aging field is the identity of the cell types that undergo the senescence gene program during the aging process. This is particularly important to better understand how senescent cell burden promotes aging-related diseases such as cancer. To answer this question, in collaboration with the Tabula Muris Consortium, we utilized next generation single-cell RNA-sequencing. We revealed that a large fraction of tissue resident macrophages expresses many of the same hallmarks of senescent cells during aging, suggesting these cells may be a key driver of inflammaging. To further understand senescent macrophage biology and their role in inflammaging, Dr. Covarrubias developed an in vitro system to grow senescent macrophages. Interestingly, senescent macrophages can be induced via treatment of quiescent macrophages with irradiation and the chemotherapeutic reagent doxorubicin; front line treatments for cancer patients. Furthermore, macrophages exposed to radial-therapies are highly resistant to cell death and have been recently shown to promote resistance to these therapies.
Highlighted Publications
Covarrubias AJ*, Perrone R*, Grozio A*, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing (Review). Nature Reviews Molecular Cell Biology. Nat Rev Mol Cell Biol. 2021 Feb;22(2):119-141. doi: 10.1038/s41580-020-00313-x. Epub 2020 Dec 22. PMID: 33353981
Covarrubias AJ, Kale A, Perrone R, Lopez-Dominguez JA, Pisco AJ, Kasler HG, Schmidt MS, Kwok R, Heckenbach I, Wiley CD, Wong HS, Gibbs E, Iyer SS, Basisty N, Wu Q, Kim IJ, Silva E, Vitangcol K, Shin KO, Lee YM, Riley R, Ben-Sahra I, Ott M, Schilling B, Scheibye-Knudsen M, Ishihara K, Quake SR, Newman J, Brenner C, Campisi J, Verdin E. The NAD hydrolase CD38 is expressed by pro-inflammatory macrophages in response to cellular senescence during aging leading to a decline in tissue NAD levels. Nature Metabolism. 2020 Nov;2(11):1265-1283. doi: 10.1038/s42255-020-00305-3. Pubmed PMID: 33199924
Chin AI, Miyahira AK, Covarrubias AJ, Teague J, Guo B, Dempsey PW, Cheng G. Toll-like receptor 3-mediated suppression of TRAMP prostate cancer shows the critical role of type-I interferons in tumor immune surveillance. Cancer Research. 2010 Apr 1; 70(7): 2595
Newman J, Covarrubias AJ, Zhao M, XinXing Y, Gut P, Ng CH, Huang Y, Haldar S, Verdin E. Ketogenic diet reduced mid-life mortality and improves memory in aging mice. Cell Metabolism. 2017. Sept 5; 26(3): 547-557
Covarrubias AJ, Aksoylar HI, Yu J, Snyder NW, Worth AJ, Iyer SS, Wang J, Ben- Sahra I, Byles V, Polynne-Stapornkul T, Espinosa EC, Lamming D, Manning BD, Zhang Y, Blair IA, Horng T. AktmTORC1 signaling regulates Acly to integrate metabolic input to control of macrophage activation. Elife. 2016 Feb 19;5