Screening Modality Test Characteristics
To inform the 2021 United States Preventive Services Task Force (USPSTF) colorectal cancer screening guideline, the US Preventive Services Task Force (USPSTF) commissioned the National Cancer Institute’s (NCI’s) Cancer
Intervention and Surveillance Modeling Network (CISNET) to provide information from comparative modeling on how estimated life-years gained, colorectal cancer cases averted, and colorectal cancer deaths averted vary by different starting and stopping ages for various screening strategies. The review also examined whether these findings varied by age, sex, or race/ethnicity. The test characteristic values used are summarized in the table adapted from the CISNET technical report.
In addition to the modeling studies, the US Preventive Services Task Force (USPSTF) commissioned a systematic review to evaluate the benefits and harms of screening for colorectal cancer in adults 40 years or older. The review also examined whether these findings varied by age, sex, or race/ethnicity.
The editorial that accompanies the 2021 guideline can be found here.
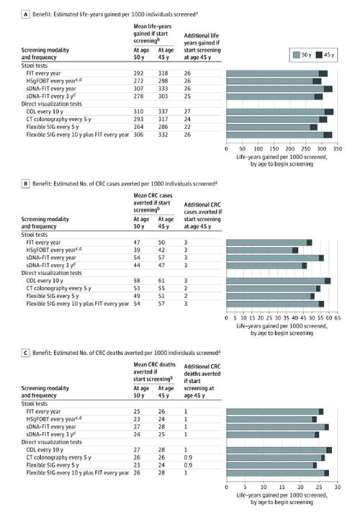
Figure legend - Click here to or on image to view PDF
Benefits of colorectal cancer screening (CRC) indicates colorectal cancer; CT, computed tomography; FIT, fecal immunochemical test (with positivity cutoff of 20 μg of hemoglobin per gram of feces); HSgFOBT, high-sensitivity guaiac fecal occult blood test; sDNA-FIT, stool DNA tests with FIT (multitarget stool DNA test); SIG, sigmoidoscopy; COL, colonoscopy.
A) Outcomes are expressed per 1000 40-year-olds who start screening at age 45 or at age 50.
B) Mean estimate across the 3 Cancer Intervention and Surveillance Modeling Network colorectal cancer models. See modeling report for additional details and model-specific estimates.
C) Because of imprecision in sensitivity and specificity, there is considerable uncertainty in model predictions for HSgFOBT strategies. See modeling report for more information.
Compared with other options for stool-based screening, these strategies do not provide an efficient balance of the benefits (life-years gained) vs harms and burden (ie, lifetime number of colonoscopies) of screening.
See modeling report for more information