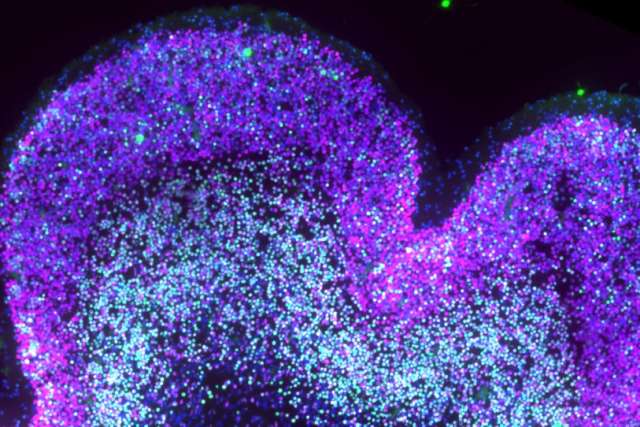
Scientists at UCLA have created one of the first cellular-resolution molecular maps detailing how Down syndrome alters human brain development before birth — a resource that resolves longstanding contradictions in the field and could lay the groundwork for future therapeutic strategies.
The study, published in Science, analyzed more than 100,000 nuclei from human prenatal neocortex samples collected across 26 pre-genotyped donors during gestational weeks 13 to 23 — the only window during which all the cortical neurons a person will carry for their entire life are generated. The findings suggest that Down syndrome disrupts the developmental sequence of that process, creating shifts that may help explain later differences in cognition, learning and sensory processing.
“There’s a new level of detail here that had never existed before,” said Luis de la Torre-Ubieta, senior author of the study and a member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA. “For the first time, we can really try to understand systematically what’s going on in the developing brain of individuals with Down syndrome.”
Filling a critical gap
The Down syndrome research field has historically focused on two areas: the adult brain and the disorder’s connection to neurodegeneration. The link is striking — the vast majority of people with Down syndrome will develop Alzheimer’s disease, typically by their 60s.
What remained largely unexamined, despite clear indicators that Down syndrome is a developmental condition — such as smaller brain volumes detectable by MRI and cognitive differences apparent as early as 6 months to one year of age — was how the condition shapes the developing brain itself.
“No one had looked at the developing human brain in Down syndrome directly using single-cell genomics,” said de la Torre-Ubieta, an assistant professor of psychiatry and biobehavioral sciences. “Mouse models and in vitro models are important tools, but don’t really give you a gold standard of what’s happening in the human brain — and actually, they have led to different results and some confusion in the field.”
These inconsistencies are due in part to differences in how mice and human brains develop, and the fact that in vitro models don’t fully represent all the cell types and tissues present in the brain.
The new study, de la Torre-Ubieta said, can serve as that gold standard resource.
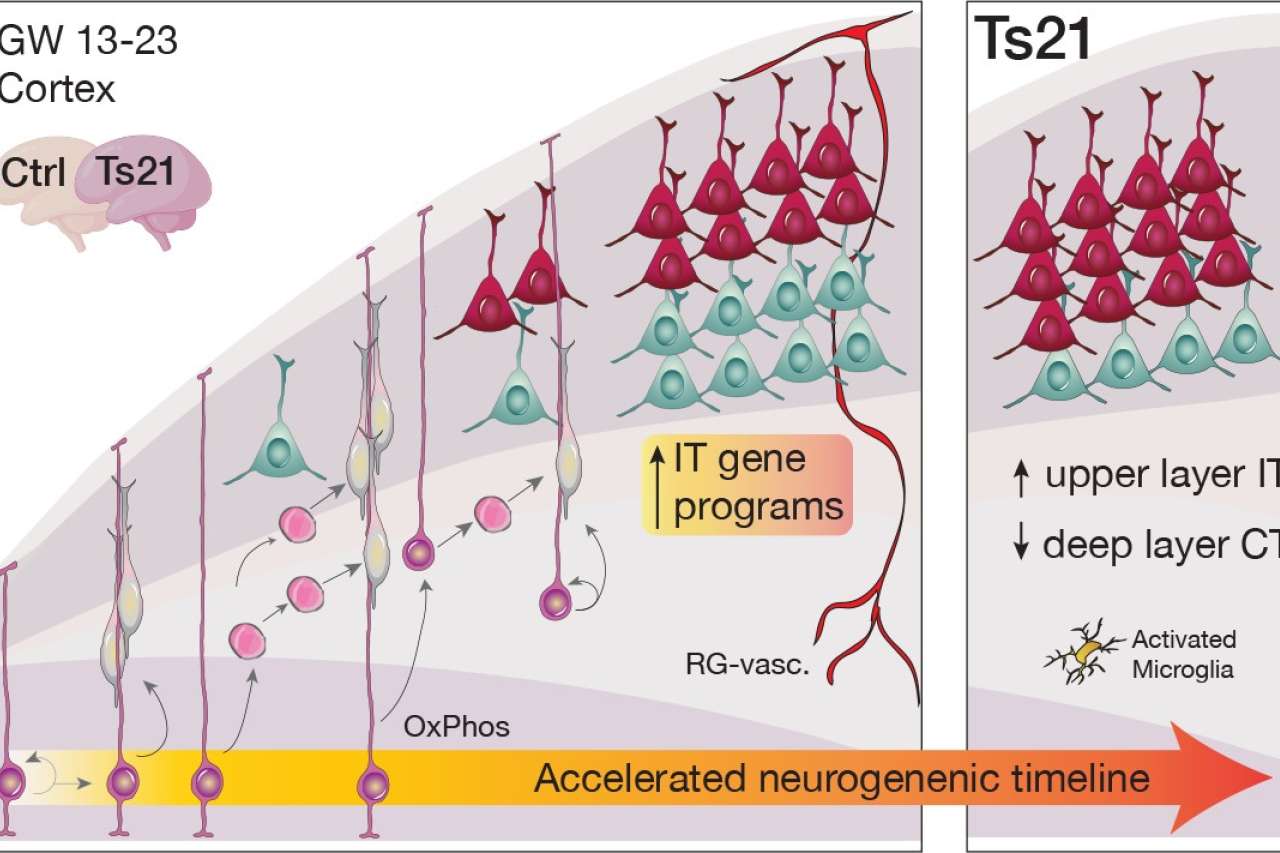
A disrupted developmental sequence and its impact on brain size
The development of the prenatal neocortex typically follows a tightly orchestrated sequence. Progenitor cells — the brain’s stem cells — must first divide repeatedly to expand their own pool, building up a sufficient base for all future neurons. Only then do they begin differentiating into neurons, starting with deep-layer cell types and progressing toward upper-layer cells in a carefully timed order.
In Down syndrome, that sequence appears to break down. The study found that progenitor cells appear to rush prematurely into neuron production, depleting their own pool and skewing the balance of neuron types generated. Specifically, the researchers observed a relative increase in upper-layer intratelencephalic neurons and a reduction in deep-layer corticothalamic neurons.
Those two cell populations play fundamentally different roles: CT neurons project outward from the cortex — connecting to brain structures and to the spinal cord to govern sensation and movement; IT neurons wire within the cortex, connecting the two hemispheres and contributing to information processing. This finding offers a new hypothesis for how early developmental changes might contribute to the cognitive profile of the condition.
The finding also offers a new answer to a longstanding question in the field: Why do people with Down syndrome tend to have smaller brains? Earlier theories centered on elevated rates of cell death. The current study found less evidence of widespread neuronal death and instead points to the depletion of the progenitor pool.
A systems-level view of a systems-level disorder
The study employed paired single-nucleus multi-omics, a technology that measures both gene expression and chromatin accessibility in the same individual cell. Chromatin accessibility reveals which regions of the genome are open and active — the enhancers and promoters that regulate gene expression — offering a layer of information beyond which genes are simply switched on or off.
By combining these two readouts, the researchers were able to reconstruct not just a snapshot of which cells are present, but the regulatory programs that guide cell fate — and how those programs are disrupted in Down syndrome. Systems-level approaches also led them to uncover alterations in cell metabolism and changes in how the vasculature interacts with the developing nervous system, both of which could speed up neuron production.
Implications beyond Down syndrome
The study’s significance extends beyond Down syndrome. The researchers specifically tested for overlap between the molecular disruptions they identified and the genetic risk signatures associated with other neurodevelopmental and neuropsychiatric conditions, including autism, epilepsy and developmental delay. They found substantial convergence, particularly in the gene-regulatory networks governing the specification of IT versus CT neurons.
“Down syndrome could be a model to understand intellectual disability and neuropsychiatric disorders more broadly,” de la Torre-Ubieta said. “Also to uncover the shared biology underlying these conditions — because the mechanisms are often still unknown.”
Two papers, one story
The publication coincides with a companion paper from researchers at the University of Wisconsin-Madison, appearing in the same issue of Science. While the UCLA study focuses on the prenatal period, the Wisconsin team examined the postnatal brain, studying Down syndrome between approximately one and five years of age.
When the two groups shared preliminary findings, they discovered striking parallels: many of the changes identified prenatally by the UCLA team appear to persist into early childhood.
Together, the two papers provide a continuous molecular view of Down syndrome brain development from mid-gestation through infancy — a resource that did not previously exist and that the researchers expect will serve as a reference for their field for years to come.
A foundation for future therapies
While the researchers are careful to emphasize that the findings do not point to a near-term clinical application, the study provides the clearest picture yet of the cellular and molecular events that distinguish the Down syndrome brain during development, and a framework for identifying future therapeutic targets.
“We are finding targets that could be actionable down the line if you generate drugs for specific pathways,” de la Torre-Ubieta said. “And you could conceive of a gene therapy based on that, to suppress the expression of particular drivers and restore development closer to its normal course.”
UCLA authors Celine K. Vuong and Alexis Weber led this work, along with Patrick Seong, Yu-Jen Chen, Jordan Peyer, Shahab Younesi, Angelo Salinda, Daniel Gomez, Gabriella Rivas, Abril Morales, Beck Shafie, Pan Zhang, Susanne Nichterwitz, Le Qi, Nolan T. Fernandez, Emily Friedman, Daniel H. Geschwind and William E. Lowry. Nana Matoba, Michael I. Love, Michael J. Gandal and Jason L. Stein contributed to this study.
The research was supported by the National Institute of Child Health and Human Development, the National Institute of Mental Health, the UCLA Broad Stem Cell Research Center, including a Rose Hills Foundation Innovator Grant and a post-doctoral training grant, the UCLA Health Jonsson Comprehensive Cancer Center and UCLA Broad Stem Cell Research Center Ablon Scholars Program, the California Institute for Regenerative Medicine and the National Institutes of Health Biomedical Big Data Training Program.