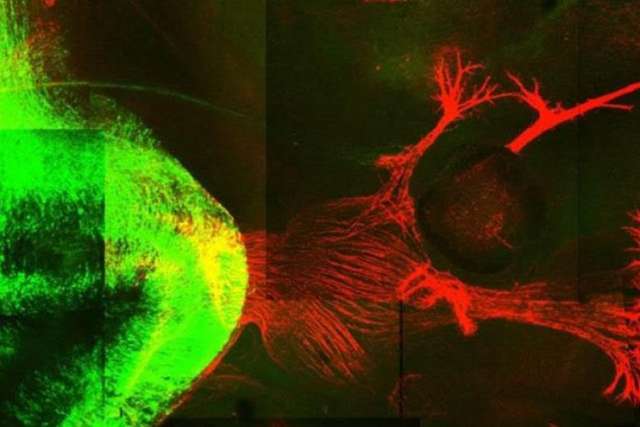
In a new study in mice, a team of researchers from UCLA, the Swiss Federal Institute of Technology, and Harvard University have uncovered a crucial component for restoring functional activity after spinal cord injury. The neuroscientists have shown that re-growing specific neurons back to their natural target regions led to recovery, while random regrowth was not effective.
In a 2018 study published in Nature, the team identified a treatment approach that triggers axons — the tiny fibers that link nerve cells and enable them to communicate — to regrow after spinal cord injury in rodents. But even as that approach successfully led to the regeneration of axons across severe spinal cord lesions, achieving functional recovery remained a significant challenge.
For the new study, published this week in Science, the team aimed to determine whether directing the regeneration of axons from specific neuronal subpopulations to their natural target regions could lead to meaningful functional restoration after spinal cord injury in mice. They first used advanced genetic analysis to identify nerve cell groups that enable walking improvement after a partial spinal cord injury.
The researchers then found that merely regenerating axons from these nerve cells across the spinal cord lesion without specific guidance had no impact on functional recovery. However, when the strategy was refined to include using chemical signals to attract and guide the regeneration of these axons to their natural target region in the lumbar spinal cord, significant improvements in walking ability were observed in a mouse model of complete spinal cord injury.
"Our study provides crucial insights into the intricacies of axon regeneration and requirements for functional recovery after spinal cord injuries,” said Michael Sofroniew, MD, PhD, professor of neurobiology at the David Geffen School of Medicine at UCLA and a senior author of the new study. “It highlights the necessity of not only regenerating axons across lesions but also of actively guiding them to reach their natural target regions to achieve meaningful neurological restoration."
The authors say understanding that re-establishing the projections of specific neuronal subpopulations to their natural target regions holds significant promise for the development of therapies aimed at restoring neurological functions in larger animals and humans. However, the researchers also acknowledge the complexity of promoting regeneration over longer distances in non-rodents, necessitating strategies with intricate spatial and temporal features. Still, they conclude that applying the principles laid out in their work “will unlock the framework to achieve meaningful repair of the injured spinal cord and may expedite repair after other forms of central nervous system injury and disease.”
Article: Recovery of walking after paralysis by regenerating characterized neurons to their natural target region, Jordan W. Squair, Marco Milano, Alexandra de Coucy, Matthieu Gautier, Michael A. Skinnider, Nicholas D. James, Newton Cho, Anna Lasne, Claudia Kathe, Thomas H. Hutson Steven Ceto, Laetitia Baud, Katia Galan, Viviana Aureli, Achilleas Laskaratos, Quentin Barraud Timothy J. Deming, Richie E. Kohman, Bernard L. Schneider, Zhigang He, Jocelyne Bloch, Michael V. Sofroniew, Gregoire Courtine, Mark A. Anderson. Science 2023 DOI: 10.1126/science.adi6412
The research team included scientists from NeuroX Institute, School of Life Sciences, Swiss Federal Institute of Technology (EPFL); the Department of Neurosurgery, Lausanne University Hospital (CHUV) and University of Lausanne (UNIL), Center for Interventional Neurotherapies (NeuroRestore); Wyss Center for Bio and Neuroengineering; Department of Clinical Neuroscience, Lausanne University Hospital (CHUV) and University of Lausanne; Departments of Bioengineering, Chemistry and Biochemistry, University of California, Los Angeles; Bertarelli Platform for Gene Therapy, Swiss Federal Institute of Technology; Brain Mind Institute, School of Life Sciences, Swiss Federal Institute of Technology; M. Kirby Neurobiology Center, Department of Neurology, Boston Children's Hospital, Harvard Medical School, Boston; Department of Neurobiology, David Geffen School of Medicine, University of California, Los Angeles.
This work was supported by the Defitech Foundation, Wings for Life, Riders4Riders, Wyss Center for Bio and Neuroengineering, Swiss National Science Foundation (PZ00P3_185728 to M.A.A. and PZ00P3_208988 to J.W.S.); the Morton Cure Paralysis Foundation (to M.A.A); the ALARME Foundation (to M.A.A. and G.C); the Dr. Miriam and Sheldon G. Adelson Medical Foundation (to M.V.S., Z.H., and T.J.D.); Wings for Life (M.A.A., M.V.S., M.A.S., and M.M); Holcim-Stiftung Foundation (to J.W.S.); and the Canadian Institutes for Health Research (to J.W.S.). We are grateful to J. Ravier and M. Burri for the illustrations and to L. Batti and I. Gantar from the Advanced Lightsheet Imaging Center (ALICe) at the Wyss Center for Bio and Neuroengineering, Geneva. Funding: This work was supported in part using the resources and services of the Gene Expression Core Facility, and the Bertarelli Platform for Gene Therapy at the School of Life Sciences of EPFL.