Research
A Culture of Research Excellence in the Department of Anesthesiology Perioperative Medicine at UCLA
The Department of Anesthesiology Perioperative Medicine at UCLA has a vibrant culture of research spanning basic, translational, and clinical disciplines. Our subject matter foci include cardiovascular biology, neuroscience, organ protection, and computational medicine, with scientists investigating critical questions of relevance to the practice of anesthesiology and the effective management of the perioperative patient. We also engage with and contribute to the larger research community at UCLA: the research conducted at the Department is strategically aligned with the research themes and initiatives of the David Geffen School of Medicine (DGSOM) and other departments, schools, and centers at UCLA.
Our Division of Molecular Medicine hosts basic and translational scientists conducting research from the scale of single molecules to whole organ physiology in vivo. All of the principal investigators in this robust research environment are funded by the National Institutes of Health (indeed, our Department is consistently among the top NIH-funded anesthesiology departments in the country) and supported by outstanding core facilities, including biochemistry, tissue culture, histology, microscopy, and in vivo imaging. Our faculty are regular speakers at prestigious scientific conferences and publish their work in high impact journals—they hold leadership positions in scientific organizations, are members of leading editorial boards, and are recipients of awards for their research accomplishments. A dedicated team of clinical research staff provides comprehensive support for studies involving patients, ranging from biomarker discovery to industry and government sponsored clinical trials.
The Department is dedicated to providing an outstanding environment for research training. We are proud of our track record of producing basic scientists who have gone on to successful careers in academia (at UCLA and other institutions) as well as in the private sector. In particular, we are committed to providing opportunities, support, and mentorship for clinician-scientists in the discipline of anesthesiology: over the past decade, several clinician-scientists who trained in our Department have gone on to lead their own extramurally-funded research programs, a model we practice currently through cultivation of a trainee pipeline including medical students, resident and fellow physicians, and junior faculty. This commitment has been further bolstered by the recent awarding of an NIH T32 training grant to our Department.
We welcome you to explore this training environment, our individual research laboratories, and the breadth of clinical research studies in the Department of Anesthesiology Perioperative Medicine at UCLA. Please reach out if you are interested to join our team!

Support Our Research
Contribute to our missions in advancing research in anesthesiology and training the next generation of physicians and scientists!

Bioinformatics & Perioperative Analytics
Data extraction, validation, and reporting; predictive analytics and risk stratification; and research in data-driven quality improvement

Clinical Research
Advancing the practical delivery of anesthesiology care and improving patient safety

T32 Research Fellowship in Anesthesiology
Designed to train the next generation of academic anesthesiologists in basic, preclinical and clinical research

Cai Lab
Interested in reactive oxygen species regulation of endothelial nitric oxide synthase (eNOS) expression and function

Civelek Lab
Studying how genetics and environment interact to shape the risk of cardiovascular and metabolic diseases. Using systems genetics, we integrate large-scale data—from gene expression and proteins to metabolites—to uncover the networks that drive disease susceptibility.
Eghbali Lab
Studying the molecular and cellular mechanisms of and developing novel therapeutics for heart disease and lung disease
Gu Lab
Using stem cell derived “miniature organs in a dish” as revolutionary platforms to understand disease progression, improve drug development, revolutionize personalized medicine, and ultimately to establish novel therapeutics for organ regeneration.
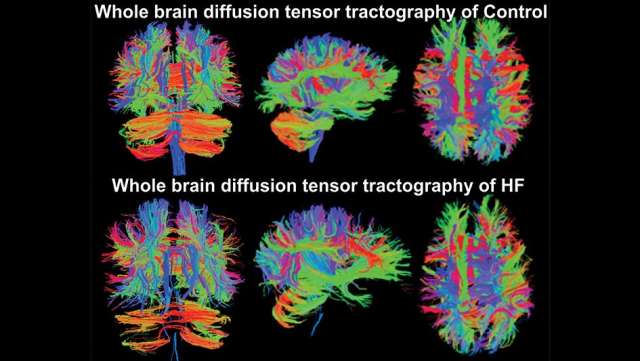
Kumar Lab
Uncovering neural processes regulating autonomic, respiratory, mood, and cognitive function

Lee Lab
Studying the biological effects of extracellular vesicles in the development of acute lung injury

Massaly Lab
Unraveling the neuronal networks and allostatic changes that might influence behavioral outcomes of pain treatment

Olcese Lab
Exploring the role of abnormal ion channel function in human diseases
Ottolia Lab
Studying mechanisms of ion transport in cardiac cells and their roles in heart excitability and contractility
Umar Lab
Investigating the molecular mechanisms of and devising novel regenerative therapies for cardiopulmonary disorders

Vondriska Lab
Discovering the basic principles of chromatin biology and investigating the causes of cardiovascular disease