UCLA researchers have developed a novel nanotechnology platform, which uses nanoparticles to deliver a selected cancer drug to the pancreatic cancer tumor environment where it induces a special form of tumor cell death known as immunogenic cell death or ICD. ICD initiated a robust anti-pancreatic cancer immune response against the primary and metastatic tumor sites.
The scientists also found that simultaneous incorporation of a special formulated small molecule, indoximod, in the nanocarrier could synergistically enhance ICD by interrupting a key immune suppressive pathway called the IDO pathway. Interference in immune suppression is important for sustaining the immune response.
The study, led by Drs. André Nel and Huan Meng, members of the UCLA California NanoSystems Institute and UCLA’s Jonsson Comprehensive Cancer Center, builds upon their prior research utilizing nanocarriers to deliver chemotherapy in pancreatic cancer.
BACKGROUND
Immunotherapy has become one of the most exciting developments in cancer therapy. Pancreatic cancer is deadly disease with a five-year survival rate of less than 6 percent. The disease is difficult to diagnose early on as symptoms typically do not appear until the cancer has undergone metastatic spread.
ICD leads to dying cancer cells expressing a protein on the tumor cell surface that provides an “eat-me” signal to antigen-presenting dendritic cells at the tumor site. This allows the dendritic cells to digest the cancer cells and present tumor antigens to cytotoxic T-lymphocytes. This response was enhanced and sustained by interfering in the immune suppressive effects of the IDO pathway.
Along with the study’s first author Jianqin Lu, Meng and Nel hypothesized that the encapsulated delivery of the chemotherapy agent, oxaliplatin, by nanocarriers could induce an enhanced ICD effect at the pancreatic cancer site. Additionally, they proposed that simultaneous incorporation of indoximod could result in a synergistic anti-pancreatic cancer immune response.
METHOD
As a proof of concept, the UCLA team used a vaccine approach to find an ICD-inducing chemotherapeutic agent by injecting dying pancreatic cancer cells under the skin of immune competent mice. The mice were then observed to see which chemo drugs will interfere with the growth of viable pancreatic cancer cells injected at a remote site. This helped to identify oxaliplatin as an ICD inducing drug that was also effective in growing tumors. Moreover, the response involved memory T-cells that provide durable immunity, which could be transferred to non-immune animals by adoptive transfer of lymphocytes. Co-delivery of indoximod further enhanced the ICD-inducing effect of oxaliplatin at the local cancer site.
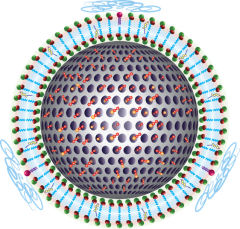
The team created nanocarriers by conjugating indoximod to a phospholipid backbone, which helps to form a “prodrug.” The prodrug is capable of self-assembly into nanovesicles or allows incorporation into a lipid bilayer that covers mesoporous silica nanoparticles. This arrangement allowed oxaliplatin to be trapped in the interior space of the silica nanoparticle pores.
Building on their previous results that use pancreatic cancer tumors growing in the pancreas of experimental animals, Lu used the contemporaneous delivery of oxaliplatin plus indoximod for delivery of the synergistic drug combination to the cancer site. The effective delivery of high drug concentrations induced a robust immune response that could eradicate the primary tumors as well as cancer metastases. Clear evidence was obtained for enhanced recruitment of cytotoxic T-lymphocytes along with disappearance of immune-suppressive regulatory T cells. This resulted in prolonged animal survival.
IMPACT
The ability to trigger an immune response in a solid tumor environment in a predictable fashion is a great advantage of ICD, which could overcome the guesswork of who may respond to immunotherapy. Moreover, the ability to use the nano-platform for dual drug delivery allows synergy between the ICD response and interference in the IDO pathway, which is not readily achievable by free roaming drugs. The dramatic impact on pancreatic cancer forecasts that similar accomplishments are achievable for other cancers, like breast and lung cancer.
Encapsulated drug delivery by nanocarriers contributes to improving drug safety, as well as targeting an immune suppressive pathway that may cause fewer side effects than seen with immune checkpoint inhibitors. Critically, the nanocarriers also contribute to threshold drug doses at the tumor site that are critical for generating a robust immune response.
“In addition to a standalone treatment platform for pancreatic cancer and other types of cancers, it is reasonable to contemplate combining the immunotherapy platform with other therapies to enhance treatment outcomes, including checkpoint inhibitors, traditional chemotherapy and radiation,” said Nel.
JOURNAL
The research is published online in Nature Communications.
AUTHORS
UCLA’s Drs. André Nel and Huan Meng are senior authors. The first author is Dr. Jianqin Lu. Other authors are Xiangsheng Liu, Yu-Pei Liao, Felix Salazar, Bingbing Sun, Wen Jiang, Chong Hyun Chang, Jinhong Jiang, Xiang Wang, and Anna Wu. Wu is also a member of UCLA’s Jonsson Comprehensive Cancer Center.
FUNDING
The research was supported by the National Institutes of Health.




