Symptoms and Causes
Disclaimer: The UCLA Health System cannot guarantee the accuracy of such information. The information is provided without warranty or guarantee of any kind. Please speak to your Physician before making any changes.
Symptoms
Kidney Symptoms
- Microscopic hematuria - blood not directly visible in urine, but present upon microscopic inspection. Often, an early and common symptom of IgAN.
- Gross hematuria - less common than microscopic hematuria, but at times the presenting symptom where blood is visible with urination (oftentimes described as tea colored)
- Proteinuria - abnormal, elevated amounts of protein in the urine
- Flank pain - pain between the upper abdomen and back, on one side of the body
- Hypertension- high blood pressure
- Edema- swelling near the eyes and in the lower extremities
- Fatigue
- Poor appetite
- Excessive thirst
- Hyperuricemia- elevated serum uric acid levels
Clinical Correlation with Respiratory Infections
- Clinical symptoms of IgAN, including gross hematuria, are strongly associated with preceding viral or bacterial infections, most often in the form of an upper respiratory tract infection.
Symptoms may vary among individuals depending on sex, age, and pre-existing conditions.
Causes
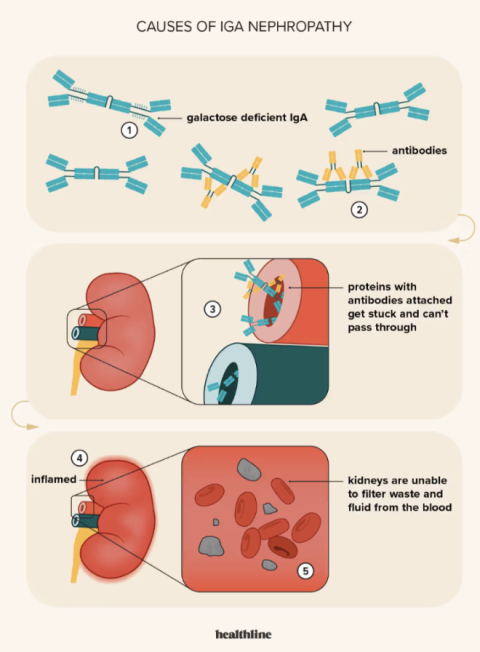
IgAN is caused by the deposition of IgA antibodies into the kidney, which are produced when the body needs to fight respiratory infections. Complex immune triggers lead to auto-antibody formation that then deposits IgA into the kidneys, leading to inflammation and eventual failure.
Genetics
Patients with microhematuria who have a family history of renal cell cancers, suspicion of Lynch syndrome, or any other known genetic renal tumor syndrome (Birt-Hogg-Dube syndrome, hereditary leiomyomatosis and renal cell cancer, hereditary papillary renal cell cancer, tuberous sclerosis complex, or von Hippel-Lindau disease) should have upper tract imaging performed regardless of other factors or their risk category. While IgAN is not strictly classified as a genetic condition, it still has a strong genetic component. Several pathogenic genes and genetic variants have been identified in IgAN through genetic testing and genome-wide association studies. For example, mutations in genes such as C1GALT1, C1GALT1C1, and GALK2 are associated with higher levels of galactose-deficient IgA1 (Gd-IgA1), a key marker of the disease. Overexpression of the cytokines TNFSF13 (APRIL) and TNFSF13B (BAFF) promotes B-cell activation, which also results in higher levels of Gd-IgA1.
Affected Populations
Patients who have a family history of IgAN or IgA vasculitis, celiac disease (gluten allergy), inflammatory bowel disease (IBD), hepatitis, cirrhosis (liver failure), or HIV (human immunodeficiency virus) are at a higher risk of developing IgA Nephropathy.
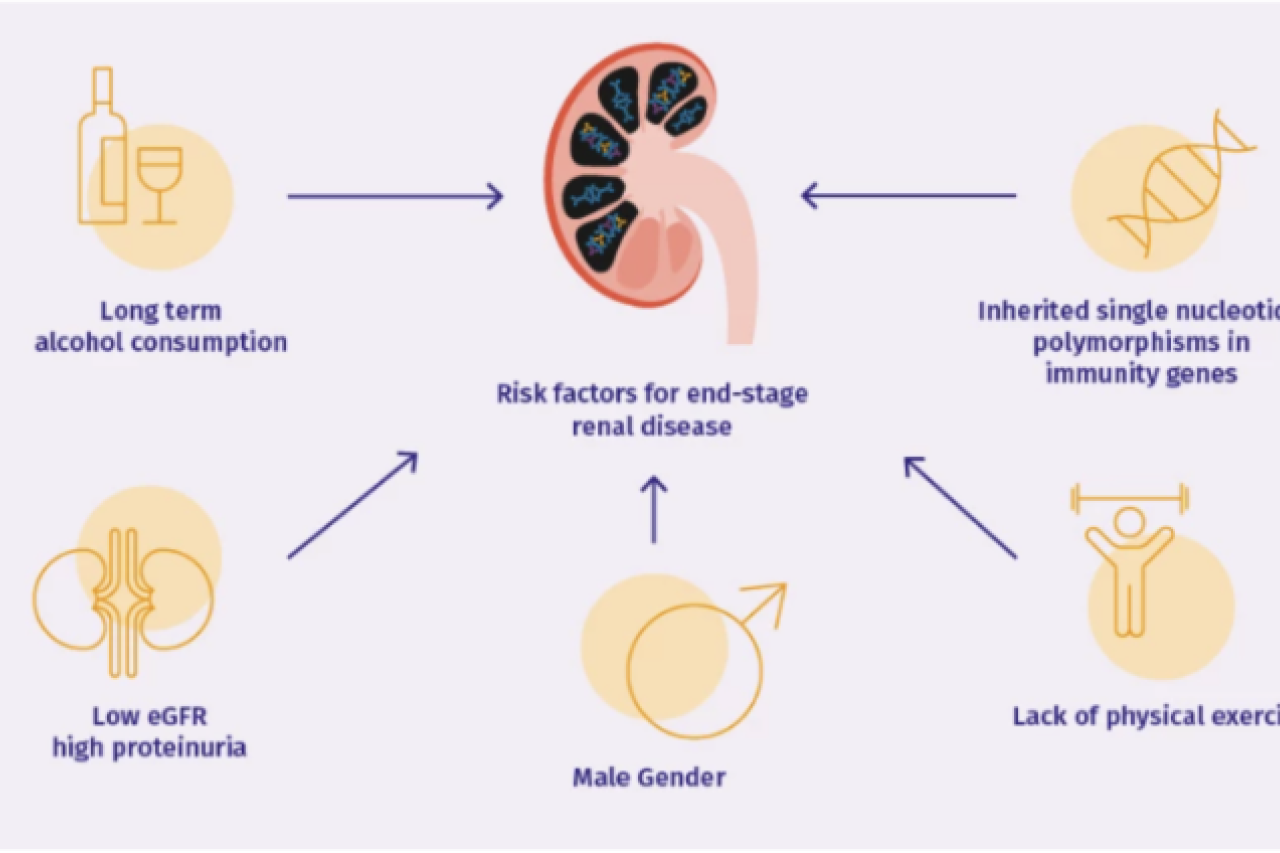
People of Asian/Pacific Islander and Hispanic ancestry have also been found to have an increased risk of developing IgAN. IgAN patients with persistent proteinuria, uncontrolled hypertension, reduced kidney function (lower eGFR), or more severe renal biopsy findings (measured by MEST-C score) at diagnosis often have an increased risk of faster eGFR decline and more rapid progression to ESRD.
Additional risk factors for end-stage renal disease: