Researchers from the UCLA School of Dentistry and the David Geffen School of Medicine at UCLA have been awarded $5 million over five years from the National Cancer Institute to develop liquid biopsy tools for testing individuals who could have lung cancer — the leading cause of cancer death in both U.S. men and women. The award, one of only six given in the nation, will support further development of the tools to improve methods for early detection of lung cancer.
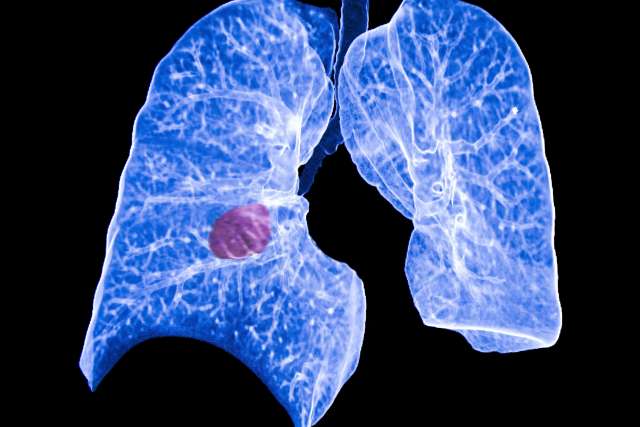
Today’s standard of care in diagnosing lung cancer involves a low-dose computed tomography scan of the lungs, evaluating whether there are what are called “indeterminate lung nodules” and following these nodules over time to assess whether they begin to look suspicious for lung cancer. The challenge of evaluating indeterminate lung nodules is differentiating between individuals with a low likelihood of lung cancer who may benefit from future scans and individuals harboring lung cancer, who may require lifesaving invasive procedures.
With a five-year lung cancer survival rate of only 18 percent, early detection is critical for those individuals at risk. There is a broad, unmet need for a noninvasive, inexpensive test to detect lung lesions that can be used in tandem with the low-dose tomography, which can reliably differentiate between indeterminate pulmonary nodules that are cancerous or benign.
The study aims to enroll at least 300 at-risk patients to test their blood and saliva using the UCLA-developed technology known as electric field-induced release and measurement, or EFIRM, liquid biopsy. The technology, which was created in the lab of Dr. David Wong, principal investigator and professor of oral biology and medicine at the UCLA School of Dentistry, takes a biological liquid sample and detects the presence of tumor mutations. Wong, who is also the dental school’s associate dean for research, and his team were able to show that the technology can detect early-stage lung cancer with greater than 90 percent similarity with traditional tissue biopsies.
“The National Cancer Institute’s award moves the needle forward for oncologists and care teams who are trying to save lives,” said Dr. Denise Aberle, principal investigator and professor of radiology at the David Geffen School of Medicine at UCLA. “In partnership with the UCLA School of Dentistry, we aim to establish a panel of 10 common biomarkers in lung cancer cases. Our ultimate goal is to use our new panel as a guide to identify at least 60 percent of lung cancer-associated biomarkers in those undetermined cases where the patient may or may not have cancer.”
Under Aberle’s guidance, blood and saliva samples will be obtained from at-risk patients being seen at the Ronald Reagan UCLA Medical Center and other UCLA Health clinics to be tested at Wong’s lab. Within a day, using the UCLA-developed technology, a diagnosis will be made as to whether the patient should undergo a biopsy. These results will be combined with those of detailed, quantitative image analyses to develop predictors of lung cancer in patients with indeterminate lung nodules.
Wei Liao, co-inventor of the technology, created EZLife Bio Inc., a biotechnology company that developed the EFIRM Reader and Plates. These prototypes and accessories enable the academic industry partnership, a requirement of the National Cancer Institute award, to translate and improve the technology for use in clinical settings. Fang Wei, adjunct associate professor at the UCLA School of Dentistry, will join the study team as an academic investigator. She is a co-inventor of the EFIRM technology.
“We are thrilled that the National Cancer Institute has recognized the potential of our revolutionary technology for the early detection of lung cancer,” said Dr. Charles Strom, medical director of EZLife Bio. “We believe that many lives can be saved by the appropriate use of EFIRM for early detection of lung cancer when it is still curable by surgical intervention.”
By tapping into the promising field of liquid biopsy and its integration with imaging, the team of researchers and clinicians would be able to place patients into specific categories, thus reducing unnecessary procedures and screening for some while identifying others who need immediate care. Liquid biopsy also provides an alternative method of testing for individuals who cannot undergo surgery because of potential complications from other conditions or other circumstances.
“Our collaborative, multidisciplinary team provides the expertise and the campuswide support needed to make significant progress in this important clinical area,” said Dr. Steve Dubinett, co-investigator and chief of pulmonary and critical care medicine in the Geffen School. “Over the next five years, we anticipate clinically validating a liquid biopsy test that can effectively and inexpensively diagnose patients who have indeterminate lung lesions in near-real time testing.”
Aberle, Dubinett and Wong are all members of the UCLA Jonsson Comprehensive Cancer Center. Dubinett is the director of the UCLA Lung Cancer Research Program, and his associate, Kostantyne Krysan, will join the research team.
Wong is co-founder of RNAmeTRIX Inc., a molecular diagnostic company. He holds equity in RNAmeTRIX Inc. and EZLife Bio USA, and serves as the company’s director and scientific advisor. The University of California also holds equity in RNAmeTRIX. Intellectual property that Wong invented and which was patented by the University of California has been licensed to RNAmeTRIX. Wong is consultant to GlaxoSmithKline, PeriRx, Wrigley and Colgate-Palmolive.