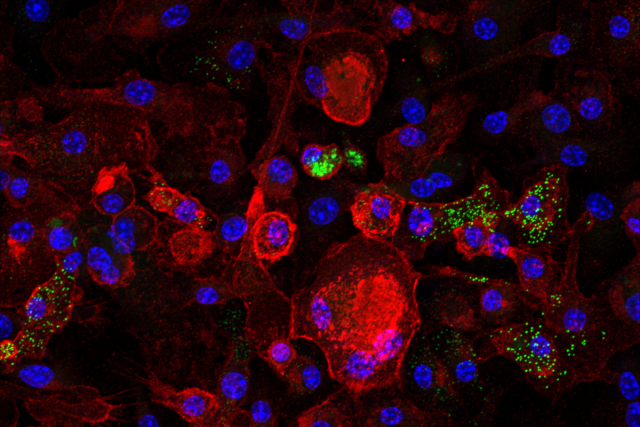
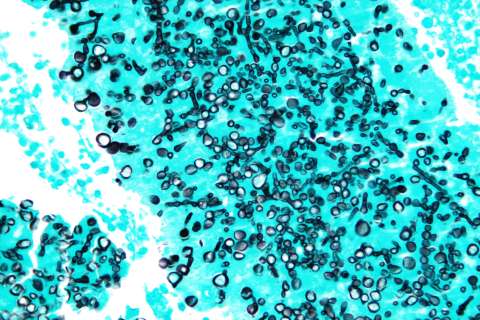
UCLA researchers have identified a rogue population of immune cells that quietly accumulates in aging tissues and in the livers of people with fatty liver disease. Clearing these cells, they found, dramatically reduced inflammation and reversed liver damage in mice — even while the animals remained on an unhealthy diet.
The study, published in Nature Aging, centers on a process called cellular senescence, a stress response in which cells stop dividing but refuse to die. Instead of quietly fading away, these so-called “zombie cells” linger in tissues and pump out a toxic cocktail of inflammatory signals.
“Senescent cells are fairly rare, but think of them like a broken-down car on the 405,” said Anthony Covarrubias, senior author of the study and a member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA. “Just one stalled car can back up traffic for miles. Now imagine five or ten of them slowly accumulating. That’s what these cells do to a tissue: even a small number causes enormous disruption.”
For years, scientists debated whether macrophages — the large immune cells that patrol every tissue in the body, engulfing debris, pathogens and dying cells — could truly become senescent. The prevailing view was that they could not. Part of the confusion stemmed from biology: macrophages naturally display some molecular markers of senescence even when healthy, making it hard to tell a genuinely dysfunctional cell from one simply doing its job.
The UCLA team resolved this by identifying a molecular signature — two proteins, p21 and TREM2, whose combination reliably flags macrophages that are genuinely senescent: no longer functional, but persistently inflaming their surrounding tissue.
Using this signature, the researchers found that the proportion of senescent macrophages in the liver surges from roughly 5% in young mice to nearly 60-80% in old ones, closely tracking with the rise of chronic liver inflammation during normal aging. But aging, it turns out, isn’t the only trigger.
Excess cholesterol, the researchers found, can also push macrophages into a senescent state. When the team exposed healthy macrophages to high levels of LDL cholesterol in the laboratory, the cells developed the hallmarks of senescence: they stopped dividing, began secreting inflammatory proteins and showed the distinctive p21-TREM2 signature.

“Physiologically, macrophages can handle cholesterol metabolism,” said Ivan Salladay-Perez, first author of the new study and a graduate student in the Covarrubias lab. “But in a chronic state, it’s pathological. And when you look at fatty liver disease, which is driven by overnutrition and too much cholesterol in the blood, that excess cholesterol appears to be a major driver of the senescent macrophage population.”
This finding raises a broader question the team is now pursuing: whether high-cholesterol and high-fat diets accelerate biological aging by driving macrophage senescence across multiple tissues — not just the liver, but potentially the brain, heart and fat.
Targeting senescent cells to reverse liver damage
To test whether clearing senescent macrophages could help, the researchers treated mice with ABT-263, a drug that selectively triggers cell death in senescent cells. The results were striking. In transgenic mice fed a high-fat, high-cholesterol diet — a model for human metabolic liver disease — the drug reduced liver weight from roughly 7% of total body weight back to a healthy 4-5% percent. Overall body weight also dropped 25% from an average of 40 grams back toward normal levels of around 30 grams. Treated livers were visibly smaller and a healthier shade of red compared to the enlarged, yellowy-fatty livers of untreated animals.
The findings suggest that clearing senescent macrophages from the liver is enough to produce significant metabolic benefits even without dietary intervention. “That’s what wowed me,” said Salladay-Perez. “Eliminating senescent cells doesn’t just slow the fatty liver — it actually reverses it.”
To determine whether the findings could be relevant to human disease, the team turned to existing patient data. Analyzing a publicly available genomic dataset from liver biopsies, they found the same senescent macrophage signature markedly elevated in diseased livers compared to healthy ones, suggesting that macrophage senescence plays a role in human chronic liver disease, not just in mice.
The finding carries particular urgency in Los Angeles, where an estimated 30-40% of residents are affected by fatty liver disease, with even higher rates in Latino communities. There are currently very few treatment options and limited non-invasive tools for diagnosing the condition at early stages.
“This is a huge public health crisis in the making,” said Covarrubias, who is also an assistant professor of microbiology, immunology and molecular genetics. “We’re seeing fatty liver disease in younger and younger people. So we’re really happy to make some inroads into understanding what’s driving it and identifying cell types we might be able to target.”
While ABT-263 is effective in mice, it’s too toxic for broad use in humans. As a next step, the research team will conduct drug-screening studies to identify new compounds that can selectively eliminate senescent macrophages without side effects.
The team is also exploring whether the same cells play a pathological role in other age-related conditions, including cancer and neurodegeneration. In the brain, for example, microglia — the macrophages of the central nervous system — may be susceptible to senescence in conditions like Alzheimer’s disease, where they are exposed to large amounts of cell debris as neurons die.
The researchers see their work as a demonstration of the geroscience hypothesis: that a single fundamental mechanism of aging — in this case, the accumulation of senescent macrophages — may underlie not one but many of the diseases that shorten and diminish human life.
“If you really understand the basic mechanisms driving inflammation with aging, you can target those same mechanisms to treat not just fatty liver disease, but atherosclerosis, Alzheimer’s and cancer,” said Salladay-Perez. “It all goes back to understanding how these cells arise in the first place.”
The study was supported by the National Institutes of Health, the Glenn Foundation for Medical Research, the American Federation for Aging Research and the UCLA-UCSD Diabetes Research Center.