Clinical Update
Newsletter Summer 2020
Advances in the Management of Cervical Cancer
Contribution by Ritu Salani, MD, MBA, director of the UCLA division of gynecologic oncology
Over the past few years, major advances have been made in the management of cervical cancer. One notable advance has been the role of immunotherapy in the treatment of recurrent disease (with PDL1 expression or CPS>1%) which received FDA approval in June 2018. With an improved understanding of the tumor biology, new areas of research are being explored, including treatment with TILs (tumor infiltrating lymphocytes) and novel therapeutics such as tisotumab vedotin.
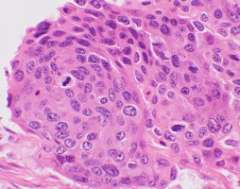
An example of squamous cell carcinoma of the cervix is shown by hematoxylin & eosin stain
At UCLA, we provide comprehensive care for women with cervical cancer from surgery and treatment (systemic therapy, radiation therapy) to survivorship care. Our expert team of gynecologic oncologists perform a wide range of surgical procedures including radical hysterectomy, sentinel lymph node assessment, lymph node debulking, and pelvic exenterations. We also have a remarkable team of radiation oncologists who are dedicated to women’s cancer care and are experts in the field of brachytherapy. Our teams have partnered to perform innovative approaches in the treatment of recurrent cervical cancer, including the ability to deliver radiation immediately post-operatively to a surgical bed for curative intent.
Most importantly we continue to search for better treatments and identify opportunities to prevent the development or recurrence of invasive cancer by actively participating in or developing clinical trials.
Clinical trials opening soon for enrollment:
- Optimizing outcomes by adding immunotherapy to chemoradiation in locally advanced disease.
- Exploring novel immunotherapy combinations in recurrent disease.
Clinical trials in development:
- Pilot study to target refractory precancerous lesions of the cervix/vulva/vagina.
- Maintenance strategies for patients with advanced/recurrent cervical cancer.
Contact our team of experts at [email protected] or our clinical trials group at [email protected].