TULSA
Find your care
Our radiologists lead the way in prostate imaging. We offer the newest techniques to better detect and stage prostate cancer. Call 310-481-7545 to find out more about prostate imaging and treatment options.
MRI-guided Transurethral Ultrasound Ablation, or TULSA, is a novel MRI-guided procedure that uses therapeutic ultrasound to effectively treat prostate cancer with minimal side effects.
How is TULSA performed?
The prostate cancer ablation treatment TULSA is a minimally invasive procedure that, under MRI guidance, delivers precise doses of therapeutic ultrasound to treat prostate cancer while sparing the healthy nerve tissue surrounding the prostate.
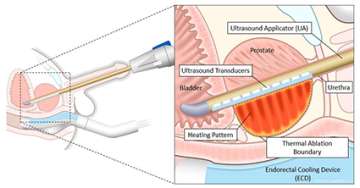
TULSA relies on a rod-shaped device that is inserted into the urethra. The novel device has 10 ultrasound-generating elements that can cover the entire prostate gland. One or more of the elements are used to send out sound waves that heat and destroy the target prostate tissue. The elements are controlled automatically by a software algorithm that can adjust the shape, direction and strength of the therapeutic ultrasound beam. The entire procedure takes place in an MRI scanner so that doctors can closely monitor treatment and assess the degree and location of heating.
"Unlike with other ultrasound systems on the market, you can monitor the ultrasound ablation process in real time and get immediate MRI feedback of the thermal dose and efficacy," said study co-author Steven S. Raman, M.D., professor of radiology and urology, and director of Prostate MR Imaging and Interventions and Prostate MR Imaging Research at the University of California at Los Angeles (UCLA).
What happens after the TULSA procedure?
Because TULSA is a minimally invasive prostate treatment, patients generally go home the same day.
What are the benefits of TULSA?
Compared with other treatment options, TULSA offers many benefits:
- Outpatient procedure
- Sophisticated imaging guidance and temperature monitoring
- Minimal recovery time
- Low rates of severe toxicity
- No bowel complications
- Preserving continence
- Preserving sexual function
- Decreased pain and discomfort
Are you a candidate for TULSA?
If you are interested in seeing if you are a candidate for TULSA, please contact our IR clinic at 310-481-7545, option 1 to arrange an appointment with one of our IR physicians.

For the clinical trial study utilizing TULSA, please review our clinical trials page