Clinical Trials
Find your care
Our radiologists lead the way in prostate imaging. We offer the newest techniques to better detect and stage prostate cancer. Call 310-481-7545 to find out more about prostate imaging and treatment options.
UCLA Prostate Imaging and Intervention is engaged in investigating new approaches to diagnosing and treating diseases and conditions.

Pivotal Study of MRI-guided Transurethral US Ablation to Treat Localized Prostate Cancer
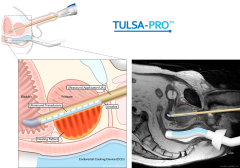
This study is not currently recruiting participants.
A prospective, multi-center, single-arm study, planned in 110 patients. The primary objective of the study is to further evaluate the safety and efficacy of a magnetic resonance imaging (MRI)-guided transurethral ultrasound therapy system (TULSA-PRO) intended to ablate prostate tissue of patients with localized, organ-confined prostate cancer.
ClinicalTrials.gov identifier: NCT02766543
Principal Investigator: Steve Raman, MD, FSAR, FSIR
RSNA Press Release: Novel MRI-Guided Ultrasound Treatment Destroys Prostate Cancer
Embosphere® PROstate Post Market Study (PROstate)
This study is not currently recruiting participants.
This is a prospective, open label post market study to evaluate the long-term safety and effectiveness of prostatic artery embolization (PAE) using Embosphere Microspheres. Up to 1,000 patients with lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) will be enrolled in this single arm post market study. All patients at sites who meet eligibility criteria will be offered participation. Long term effectiveness of PAE on LUTS will be evaluated by International Prostate Symptom Score (IPSS) at baseline, 3 months, 12 months, 24 months and 36 months. Safety will be assessed by evaluating treatment related adverse events at the same time points, plus at 4 weeks following embolization. Erectile function will be assessed at baseline and 12 months by Sexual Health Inventory for Men (SHIM) score. Additional treatments for refractory or recurrent LUTS due to BPH post prostatic artery embolization will also be recorded.
ClinicalTrials.gov identifier: NCT03527589
Principal Investigator: Justin McWilliams, MD, FSIR
Focal MR-Guided Focused Ultrasound Treatment of Localized Intermediate Risk Prostate Lesions
This study is not currently recruiting participants.
The hypothesis of this study is that focal treatment with ExAblate MRgFUS has the potential to be an effective non-invasive treatment for intermediate risk, organ-confined prostate lesions, with a low incidence of morbidity. The study hypothesis will be tested by measuring treatment-related safety and initial effectiveness parameters in the ExAblate MRgFUS treated patients, as described above.
ClinicalTrials.gov identifier: NCT01657942
Principal Investigator: Steve Raman, MD, FSAR, FSIR