Case: Lobular Carcinoma in Situ (LCIS)
By: Amy Zhang, MD and Reza Fardanesh, MD
Introduction, Demographics, Clinical Presentation
Lobular carcinoma in situ (LCIS) is a noninvasive lesion of the breast considered to be a nonobligate precursor of invasive lobular carcinoma1. The pathologic definition is proliferation of atypical cells filling and distending at least half of the acini in the lobular unit2. In patients diagnosed with primary LCIS, there is a 7.1% incidence of invasive breast cancer at 10 years, with nearly identical incidence in ipsilateral versus contralateral breasts3.
LCIS has been described as clinically occult. It is found to be multicentric in most cases and is often bilateral4. Typically, an incidental finding on a biopsy performed for a separate reason, studies report the incidence of LCIS as 0.8-3.8% in open surgical biopsies and 0.02-3.3% of image-guided core needle biopsies. There is a much lower incidence demonstrated in population-based data2.
LCIS is seen most often in premenopausal women (mean age of 49 years)5. In postmenopausal women, long-term HRT use is associated with the risk of developing LCIS6.
Imaging Features
The imaging features of LCIS are nonspecific. On mammography, LCIS is most frequently occult5,7. When present, the most common mammographic finding is microcalcifications which may be of variable morphology and distribution, most often grouped and amorphous4. Of note, in many cases microcalcifications are found in tissue adjacent to the focus of LCIS instead of within the LCIS itself5.
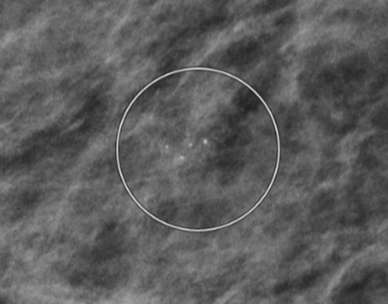
On sonography, LCIS may appear as oval, hypoechoic masses with circumscribed or microlobulated margins Posterior enhancement is common. In 32% of cases, LCIS is seen as vague shadowing or distortion without a discrete mass8.
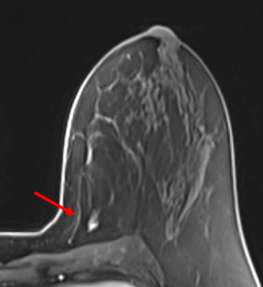
When LCIS is seen on MRI, the most common finding is non-mass enhancement. Most of the non-mass enhancement show persistent enhancement on delayed phase4.
Pathology
In LCIS, cells typically show dyscohesion with loss of E-cadherin expression. Pagetoid spread of cells between the epithelial and myoepithelial layers is another common pathologic feature9.
Besides classic LCIS, less common variants of LCIS have been recognized. Pleomorphic LCIS demonstrates large, pleomorphic nuclei. Florid LCIS involves marked distension of ducts or terminal duct lobular units. Both pleomorphic and florid LCIS subtypes are often associated with calcifications and comedonecrosis10.
Treatment
The management of LCIS is currently evolving and varies across institutions, with some recommending surgical excision and others surveillance11,12,13. This is related to the variable upstaging rate of LCIS to invasive breast cancer or to DCIS, which ranges from 2% to 25%11.
According to 2023 NCCN guidelines, a core biopsy showing classic LCIS can be managed without surgical excision, which should be considered on a case-by-case basis. If excision is not performed, the patient is recommended to undergo close follow-up with mammogram and breast MRI screening11.
The role of screening MRI in patients with LCIS is not well established. One retrospective study demonstrated additional work-up in 16.7% of patients after excision of LCIS who underwent screening MRI, with PPV of only 20%14.
Chemoprevention with selective estrogen receptor modulars or aromatase inhibitors can reduce the risk of breast cancer in patients with LCIS15.
References
- Murray MP, Luedtke C, Liberman L, Nehhozina T, Akram M, Brogi E. "Classic Lobular Carcinoma in Situ and Atypical Lobular Hyperplasia at Percutaneous Breast Core Biopsy: Outcomes of Prospective Excision." Cancer. 2013 Mar 1;119(5):1073-9. DOI: 10.1002/cncr.27841. Epub 2012 Nov 6. PMID: 23132235.
- Oppong BA, King TA. "Recommendations for Women with Lobular Carcinoma in Situ (LCIS)." Oncology (Williston Park). 2011 Oct;25(11):1051-6, 1058. PMID: 22106556.
- Chuba PJ, Hamre MR, Yap J, Severson RK, Lucas D, Shamsa F, Aref A. "Bilateral Risk for Subsequent Breast Cancer After Lobular Carcinoma-in-Situ: Analysis of Surveillance, Epidemiology, and End Results Data." J Clin Oncol. 2005 Aug 20;23(24):5534-41. DOI: 10.1200/JCO.2005.04.038. PMID: 16110014.
- Scoggins M, Krishnamurthy S, Santiago L, Yang W. "Lobular Carcinoma in Situ of the Breast: Clinical, Radiological, and Pathological Correlation." Acad Radiol. 2013 Apr;20(4):463-70. DOI: 10.1016/j.acra.2012.08.020. Erratum in: Acad Radiol. 2013 Jun;20(6):790. PMID: 23498988.
- Beute BJ, Kalisher L, Hutter RV. "Lobular Carcinoma in Situ of the Breast: Clinical, Pathologic, and Mammographic Features." AJR Am J Roentgenol. 1991 Aug;157(2):257-65. DOI: 10.2214/ajr.157.2.1853802. PMID: 1853802.
- Timbres J, Kohut K, Caneppele M, Troy M, Schmidt MK, Roylance R, Sawyer E. "DCIS and LCIS: Are the Risk Factors for Developing In Situ Breast Cancer Different?" Cancers (Basel). 2023 Sep 2;15(17):4397. DOI: 10.3390/cancers15174397. PMID: 37686673; PMCID: PMC10486708.
- Choi BB, Kim SH, Park CS, Cha ES, Lee AW. "Radiologic Findings of Lobular Carcinoma in Situ: Mammography and Ultrasonography." J Clin Ultrasound. 2011 Feb;39(2):59-63. DOI: 10.1002/jcu.20772. Epub 2010 Dec 7. PMID: 21213330.
- Ferré R, Omeroglu A, Mesurolle B. "Sonographic Appearance of Lesions Diagnosed as Lobular Neoplasia at Sonographically Guided Biopsies." AJR Am J Roentgenol. 2017 Mar;208(3):669-675. DOI: 10.2214/AJR.15.15056. Epub 2017 Jan 11. PMID: 28075608.
- Shaaban AM. "Why is LCIS Important—Pathological Review." Curr Breast Cancer Rep 13, 132–140 (2021). Published online April 7, 2021. DOI:https://doi.org/10.1007/s12609-021-00415-1
- Schnitt SJ, Brogi E, Chen YY, King TA, Lakhani SR. "American Registry of Pathology Expert Opinions: The Spectrum of Lobular Carcinoma in Situ: Diagnostic Features and Clinical Implications." Ann Diagn Pathol. 2020 Apr;45:151481. DOI: 10.1016/j.anndiagpath.2020.151481. Epub 2020 Feb 15. PMID: 32120324; PMCID: PMC7401835.
- Jani C, Lotz M, Keates S, Gupta Y, Walker A, Al Omari O, Parvez A, Patel D, Gnata M, Perry J, Khorashadi L, Weissmann L, Pories SE. "Management of Lobular Neoplasia Diagnosed by Core Biopsy." Breast J. 2023 Apr 18;2023:8185446. DOI: 10.1155/2023/8185446. PMID: 37114120; PMCID: PMC10129432.
- Laws A, Katlin F, Nakhlis F, Chikarmane SA, Schnitt SJ, King TA. "Atypical Lobular Hyperplasia and Classic Lobular Carcinoma In Situ Can Be Safely Managed Without Surgical Excision." Ann Surg Oncol. 2022 Mar;29(3):1660-1667. DOI: 10.1245/s10434-021-10827-z. Epub 2021 Sep 23. PMID: 34554341.
- Karimi Z, Phillips J, Brook A, Baker G, Guzman Y, Mehta TS. "Upgrade Rates of Pure, Radiology-Pathology Concordant Lobular Neoplasia Diagnosed on Breast Core Needle Biopsy: Is Surgical Excision Warranted?" Acad Radiol. 2022 Jul;29(7):1029-1038. DOI: 10.1016/j.acra.2021.09.009. Epub 2021 Oct 23. PMID: 34702673.
- Schwartz T, Cyr A, Margenthaler J. "Screening Breast Magnetic Resonance Imaging in Women with Atypia or Lobular Carcinoma in Situ." J Surg Res. 2015 Feb;193(2):519-22. DOI: 10.1016/j.jss.2014.08.059. Epub 2014 Sep 4. PMID: 25277350.
- Trivedi MS, Coe AM, Vanegas A, Kukafka R, Crew KD. "Chemoprevention Uptake among Women with Atypical Hyperplasia and Lobular and Ductal Carcinoma In Situ." Cancer Prev Res (Phila). 2017 Aug;10(8):434-441. DOI: 10.1158/1940-6207.CAPR-17-0100. Epub 2017 Jun 13. PMID: 28611039; PMCID: PMC5540777.